8-K
false000153117700015311772026-03-042026-03-04
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
|
Date of Report (Date of earliest event reported): March 04, 2026 |
Sight Sciences, Inc.
(Exact name of Registrant as Specified in Its Charter)
|
|
|
|
|
Delaware |
001-40587 |
80-0625749 |
(State or Other Jurisdiction
of Incorporation) |
(Commission File Number) |
(IRS Employer
Identification No.) |
|
|
|
|
|
4040 Campbell Avenue Suite 100 |
|
Menlo Park, California |
|
94025 |
(Address of Principal Executive Offices) |
|
(Zip Code) |
|
Registrant’s Telephone Number, Including Area Code: 877 266-1144 |
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
☐Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
☐Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
☐Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
☐Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
|
|
Title of each class
|
|
Trading
Symbol(s) |
|
Name of each exchange on which registered
|
Common Stock, $0.001 par value per share |
|
SGHT |
|
The Nasdaq Global Select Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ☒
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 2.02 Results of Operations and Financial Condition
On March 4, 2026, Sight Sciences, Inc. (the “Company”) issued a press release announcing its financial results for the year and quarter ended December 31, 2025. A copy of the press release is furnished as Exhibit 99.1 to this Current Report on Form 8-K (this “Current Report").*
Item 7.01 Regulation FD Disclosure
On March 4, 2026, the Company posted an investor presentation to its website at https://investors.sightsciences.com/. The Company expects to use the investor presentation, in whole or in part, and possibly with modifications, in connection with presentations to investors, analysts, and others. A copy of the investor presentation is furnished as Exhibit 99.2 to this Current Report.*
Item 9.01 Financial Statements and Exhibits
(d) Exhibits
|
|
* |
The information in Item 2.02, Item 7.01, Exhibit 99.1 and Exhibit 99.2 of this Current Report shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that Section, nor shall it be deemed to be incorporated by reference into any filing of the Company under the Securities Act of 1933, as amended, or the Exchange Act, except as expressly set forth by specific reference in such filing. |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
|
|
|
|
|
|
|
Sight Sciences, Inc. |
|
|
|
|
Date: |
March 4, 2026 |
By: |
/s/ James Rodberg |
|
|
|
James Rodberg
Chief Financial Officer
(Principal Financial and Accounting Officer) |
EX-99.1

Exhibit 99.1
Sight Sciences Reports Fourth Quarter and Full Year 2025 Financial Results and
Initiates Full Year 2026 Financial Guidance
MENLO PARK, Calif., March 4, 2026 (GLOBE NEWSWIRE) -- Sight Sciences, Inc. (Nasdaq: SGHT) ("Sight Sciences" or the "Company"), an eyecare technology company focused on developing and commercializing innovative, interventional technologies intended to transform care and improve patients’ lives, today reported financial results for the fourth quarter and full year ended December 31, 2025 and initiated financial guidance for full year 2026.
Recent Financial Highlights
•Generated fourth quarter 2025 total revenue of $20.4 million, an increase of 7% compared to the same period in the prior year, and full year 2025 total revenue of $77.4 million, a decrease of 3% compared to full year 2024.
•Achieved total gross margin of 87% in the fourth quarter of 2025 compared to 87% in the same period in the prior year, and full year 2025 total gross margin of 86% compared to 85% in full year 2024.
•Achieved a full year 2025 operating expense reduction of 13% compared to full year 2024 and a full year 2025 non-GAAP adjusted operating expense1,2 reduction of 13% compared to full year 2024.
•Reduced cash usage to $0.4 million in the fourth quarter 2025, reflecting continued operating discipline. Cash and cash equivalents totaled $92.0 million as of December 31, 2025.
Management Commentary
“We closed the year with a solid fourth quarter, where we returned to growth in Interventional Glaucoma, demonstrated encouraging commercial traction following significant reimbursement milestones in Interventional Dry Eye, and continued our disciplined expense and cash management,” said Paul Badawi, Co-Founder and CEO of Sight Sciences. “These achievements underscore the strength of our interventional technologies and commercial infrastructure in the large and growing glaucoma and dry eye markets. In 2026, we remain focused on building a leading interventional eye care company and leveraging the complementary nature of our two interventional businesses to drive synergistic growth with a pathway toward cashflow breakeven.”
Fourth Quarter 2025 Financial Results
Revenue for the fourth quarter of 2025 was $20.4 million, an increase of 7% compared to the same period in the prior year. Interventional Glaucoma revenue was $19.7 million, an increase of 5% compared to the same period in the prior year. This improvement was primarily driven by an increase in both ordering accounts and average selling prices. Interventional Dry Eye revenue was $0.7 million, an increase from $0.3 million in the same period in the prior year, primarily due to increased average selling prices.
Gross profit for the fourth quarter of 2025 was $17.8 million compared to $16.6 million in the same period in the prior year. Gross margin for the fourth quarter of 2025 was 87%, compared to 87% in the same period in the prior year. Interventional Glaucoma gross margin in the fourth quarter of 2025 increased to 88%, compared to 87% in the same period in the prior year, primarily due to higher average selling prices and product mix, slightly offset by tariff costs. Interventional Dry Eye gross margin in the fourth quarter of 2025 increased to 68%, from 51% in the same period in the prior year, primarily due to higher average selling prices.
Total operating expenses were $21.5 million in the fourth quarter of 2025, representing a 25% decrease compared to $28.5 million in the same period in the prior year, primarily due to lower personnel-related expenses and stock-based compensation. Research and development expenses were $2.4 million in the fourth quarter of 2025 compared to $4.3 million in the same period in the prior year, representing a 43% decrease. Selling, general, and administrative expenses were $19.0 million in the fourth quarter of 2025, compared to $24.2 million in the same period in the prior

year, representing a 21% decrease. Adjusted operating expenses1,2 were $18.9 million in the fourth quarter of 2025, down from $24.4 million in the same period in the prior year, representing a 23% decrease.
Net loss was $4.2 million, or a loss of $0.08 per share, in the fourth quarter of 2025, compared to a net loss of $11.8 million, or a loss of $0.23 per share, in the same period in the prior year.
Full Year 2025 Financial Results
Revenue for full year 2025 was $77.4 million, a decrease of 3% compared to full year 2024. Interventional Glaucoma revenue was $75.7 million for full year 2025, flat compared to full year 2024. Interventional Dry Eye revenue was $1.6 million for full year 2025, compared to $4.0 million in full year 2024.
Gross profit for full year 2025 was $66.7 million compared to $68.3 million in full year 2024 primarily due to lower sales volume and product mix. Gross margin for full year 2025 was 86% compared to 85% in full year 2024. Interventional Glaucoma gross margin in full year 2025 was 87% compared to 88% in full year 2024, the decrease was primarily due to higher tariff costs. Interventional Dry Eye gross margin in full year 2025 increased to 59%, from 46% in full year 2024, primarily due to higher average selling prices.
Total operating expenses were $103.8 million in full year 2025, representing a 13% decrease compared to $118.8 million in full year 2024. The decrease was primarily due to lower personnel-related expenses, legal expenses and stock-based compensation. Research and development expenses were $14.6 million in full year 2025 compared to $18.0 million in full year 2024, representing a 19% decrease. Selling, general, and administrative expenses were $89.2 million in full year 2025, compared to $100.8 million in full year 2024, representing a 12% decrease. Adjusted operating expenses1,2 were $87.8 million in full year 2025, down from $101.3 million in full year 2024, representing a 13% decrease.
Net loss was $38.4 million, or a loss of $0.74 per share, for full year 2025, compared to a loss of $51.5 million, or a loss of $1.03 per share, for full year 2024.
Cash and cash equivalents totaled $92.0 million and total long-term debt was $40.0 million (before debt discount and amortized debt issuance costs) as of December 31, 2025, compared to $120.4 million and $40.0 million, respectively, as of December 31, 2024. Cash used in full year 2025 totaled $28.4 million, compared to $17.8 million in full year 2024.
2026 Financial Guidance
Sight Sciences expects its revenue for full year 2026 to range from $82 million to $88 million, representing growth of 6% to 14% compared to full year 2025. This revenue guidance includes Interventional Glaucoma segment revenue of $77 million to $81 million, representing growth of 2% to 7% and Interventional Dry Eye segment revenue of $5 million to $7 million, compared to $1.6 million in 2025.
The Company expects adjusted operating expenses1,3 for full year 2026 to range from $93 million to $96 million, representing an increase of 6% to 9% compared to 2025. This increase is primarily due to targeted investments in both business segments, including expanded market access efforts and additional commercial resources to scale the reimbursed dry eye market and the pseudophakic standalone glaucoma opportunity.
1 “Adjusted operating expenses” is a financial measure not prepared in accordance with generally accepted accounting principles in the United States (“GAAP”, and therefore such a measure, is a “non-GAAP financial measure”), and is calculated as operating expenses less stock-based compensation expense, depreciation and amortization, restructuring costs, and other one-time costs. Please see the “Non-GAAP Financial Measures” section below for additional information.
2 A reconciliation of the non-GAAP financial measures to the most directly comparable GAAP financial measures has been provided in the table titled "Non-GAAP to GAAP Reconciliation" attached to this press release.

3 Consistent with Securities and Exchange Commission (“SEC”) regulations, the Company has not provided a reconciliation of forward-looking non-GAAP financial measures to the most directly comparable GAAP financial measures in reliance on the “unreasonable efforts” exception set forth in the applicable regulations, because there is substantial uncertainty associated with predicting any future adjustments that may be made to the Company’s GAAP financial measures in calculating the non-GAAP financial measures.
Non-GAAP Financial Measures
Adjusted operating expenses, a non-GAAP financial measure, is presented in this press release to provide information that may assist investors in understanding the Company's financial and operating results. The Company believes this non-GAAP financial measure is an important performance indicator because it excludes items that are unrelated to, and may not be indicative of, the Company's core financial and operating results. This non-GAAP financial measure, as calculated, may not necessarily be comparable to similarly titled measures of other companies and may not be an appropriate measure for comparing the performance of other companies relative to the Company. This non-GAAP financial measure is not intended to represent, and should not be considered to be a more meaningful measure than, or an alternative to, measures of operating performance as determined in accordance with GAAP. To the extent the Company utilizes such non-GAAP financial measure in the future, it expects to calculate it using a consistent method from period to period.
Conference Call
Sight Sciences' management team will host a conference call today, March 4, 2026, beginning at 1:30 p.m. Pacific Time / 4:30 p.m. Eastern Time. Investors interested in listening to the conference call may do so by accessing a live and archived webcast of the event at www.sightsciences.com, on the Investors page in the News & Events section.
About Sight Sciences
Sight Sciences is an eyecare technology company focused on developing and commercializing innovative and interventional solutions intended to transform care and improve patients’ lives. Using minimally invasive or non-invasive approaches to target the underlying causes of the world’s most prevalent eye diseases, Sight Sciences seeks to create more effective treatment paradigms that enhance patient care and supplant conventional outdated approaches. The Company’s OMNI® Surgical System and OMNI® Edge Surgical System are implant-free, minimally invasive glaucoma surgery technologies indicated in the United States to reduce intraocular pressure in adult patients with primary open-angle glaucoma. The OMNI Surgical System is CE Marked for the catheterization and transluminal viscodilation of Schlemm’s canal and cutting of the trabecular meshwork to reduce intraocular pressure in adult patients with open-angle glaucoma. Glaucoma is the world’s leading cause of irreversible blindness. The SION® Surgical System is a bladeless, manually operated device used in ophthalmic surgical procedures to excise trabecular meshwork. The Company’s TearCare® System is 510(k) cleared in the United States for the application of localized heat therapy in adult patients with evaporative dry eye disease due to meibomian gland disease (MGD), enabling clearance of gland obstructions by physicians to address the leading cause of dry eye disease. Visit www.sightsciences.com for more information.
Sight Sciences, the Sight Sciences logo, TearCare, and SmartLids are trademarks of Sight Sciences registered in the United States. OMNI and SION are trademarks of Sight Sciences registered in the United States, European Union and other territories.
© 2026 Sight Sciences. All rights reserved.
Forward-Looking Statements
This press release, together with other statements and information publicly disseminated by the Company, contains certain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. The Company intends such forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in the Private Securities Litigation Reform Act of 1995 and includes this statement for purposes of complying with these safe harbor

provisions. Any statements made in this press release or during the earnings call that are not statements of historical fact, including statements about our beliefs and expectations, are forward-looking statements and should be evaluated as such. Forward-looking statements include, but are not limited to, statements concerning our focus on advancing our strategic initiatives, including our focus in 2026 on building a leading interventional eye care company and leveraging the complementary nature of our two interventional businesses to drive synergistic growth with a pathway toward cashflow breakeven; 2026 revenue guidance; and 2026 adjusted operating expenses guidance, including primary factors impacting this guidance.
These statements often include words such as "anticipate," "expect," “suggests,” “plan,” “believe,” “intend,” “estimates,” “targets,” “projects,” “should,” “could,” “would,” “may,” “will,” “forecast” and other similar expressions. We base these forward-looking statements on our current expectations, plans and assumptions we have made in light of our experience in the industry, as well as our perceptions of historical trends, current conditions, expected future developments and other factors we believe are appropriate under the circumstances at such time. Although we believe these forward-looking statements are based on reasonable assumptions at the time they are made, you should be aware that many factors could affect our business, results of operations and financial condition, including without limitation changes to reimbursement coverage or payment decisions or reimbursement rates for our products; pricing pressure or changes in market share resulting from the evolving competitive landscape; the impact of tariffs on our products and the medical device industry generally; and disruptions to or increased costs associated with our supply chain, including as a result of having a limited number of suppliers. Should our underlying assumptions prove incorrect, actual results may differ materially from those expressed in the forward-looking statements. These statements are not guarantees of future performance or results. These forward-looking statements are subject to and involve numerous risks, uncertainties and assumptions, including those discussed under the caption “Risk Factors” in our filings with the SEC, as may be updated from time to time in subsequent filings, and you should not place undue reliance on these statements. These cautionary statements are made only as of the date of this press release. We undertake no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by applicable law.
Investor contact:
Philip Taylor
Gilmartin Group
415.937.5406
Investor.Relations@Sightsciences.com
Media contact:
pr@SightSciences.com

SIGHT SCIENCES, INC.
Consolidated Balance Sheets (Unaudited)
(in thousands, except share and per share data)
|
|
|
|
|
|
|
|
|
|
|
December 31, |
|
|
December 31, |
|
|
|
2025 |
|
|
2024 |
|
Assets |
|
|
|
|
|
|
Current assets: |
|
|
|
|
|
|
Cash and cash equivalents |
|
$ |
91,965 |
|
|
$ |
120,357 |
|
Accounts receivable, net of allowance for credit losses of $234 and $689 at December 31, 2025 and 2024, respectively |
|
|
9,745 |
|
|
|
10,786 |
|
Inventory, net |
|
|
7,767 |
|
|
|
6,325 |
|
Prepaid expenses and other current assets |
|
|
3,257 |
|
|
|
2,306 |
|
Total current assets |
|
|
112,734 |
|
|
|
139,774 |
|
Property and equipment, net |
|
|
1,610 |
|
|
|
1,580 |
|
Operating lease right-of-use assets |
|
|
438 |
|
|
|
935 |
|
Other noncurrent assets |
|
|
518 |
|
|
|
550 |
|
Total assets |
|
$ |
115,300 |
|
|
$ |
142,839 |
|
Liabilities and stockholders’ equity |
|
|
|
|
|
|
Current liabilities: |
|
|
|
|
|
|
Accounts payable |
|
$ |
1,343 |
|
|
$ |
1,691 |
|
Accrued compensation |
|
|
6,074 |
|
|
|
9,680 |
|
Accrued and other current liabilities |
|
|
3,610 |
|
|
|
4,097 |
|
Total current liabilities |
|
|
11,027 |
|
|
|
15,468 |
|
Long-term debt |
|
|
40,300 |
|
|
|
39,356 |
|
Other noncurrent liabilities |
|
|
31 |
|
|
|
492 |
|
Total liabilities |
|
|
51,358 |
|
|
|
55,316 |
|
Commitments and contingencies |
|
|
|
|
|
|
Stockholders’ equity: |
|
|
|
|
|
|
Preferred stock, par value $0.001 per share; 10,000,000 shares authorized; no shares issued and outstanding as of December 31, 2025 and 2024, respectively |
|
|
— |
|
|
|
— |
|
Common stock, par value $0.001 per share; 200,000,000 shares authorized; 53,493,711 and 50,937,999 shares issued and outstanding as of December 31, 2025 and 2024, respectively |
|
|
54 |
|
|
|
51 |
|
Additional paid-in-capital |
|
|
448,611 |
|
|
|
433,769 |
|
Accumulated deficit |
|
|
(384,723 |
) |
|
|
(346,297 |
) |
Total stockholders’ equity |
|
|
63,942 |
|
|
|
87,523 |
|
Total liabilities and stockholders’ equity |
|
$ |
115,300 |
|
|
$ |
142,839 |
|

SIGHT SCIENCES, INC.
Consolidated Statements of Operations and Comprehensive Loss (Unaudited)
(in thousands, except share and per share data)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended
December 31, |
|
|
Years Ended
December 31, |
|
|
|
2025 |
|
|
2024 |
|
|
2025 |
|
|
2024 |
|
Revenue |
|
$ |
20,385 |
|
|
$ |
19,074 |
|
|
$ |
77,363 |
|
|
$ |
79,866 |
|
Cost of goods sold |
|
|
2,597 |
|
|
|
2,513 |
|
|
|
10,697 |
|
|
|
11,581 |
|
Gross profit |
|
|
17,788 |
|
|
|
16,561 |
|
|
|
66,666 |
|
|
|
68,285 |
|
Operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
Research and development |
|
|
2,437 |
|
|
|
4,293 |
|
|
|
14,606 |
|
|
|
17,991 |
|
Selling, general and administrative |
|
|
19,020 |
|
|
|
24,197 |
|
|
|
89,159 |
|
|
|
100,826 |
|
Total operating expenses |
|
|
21,457 |
|
|
|
28,490 |
|
|
|
103,765 |
|
|
|
118,817 |
|
Loss from operations |
|
|
(3,669 |
) |
|
|
(11,929 |
) |
|
|
(37,099 |
) |
|
|
(50,532 |
) |
Investment income |
|
|
834 |
|
|
|
1,289 |
|
|
|
3,973 |
|
|
|
5,917 |
|
Interest expense |
|
|
(1,289 |
) |
|
|
(1,161 |
) |
|
|
(5,142 |
) |
|
|
(4,662 |
) |
Loss on debt extinguishment |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(1,962 |
) |
Other expense, net |
|
|
(30 |
) |
|
|
(7 |
) |
|
|
(148 |
) |
|
|
(32 |
) |
Loss before income taxes |
|
|
(4,154 |
) |
|
|
(11,808 |
) |
|
|
(38,416 |
) |
|
|
(51,271 |
) |
Provision for income taxes |
|
|
8 |
|
|
|
38 |
|
|
|
10 |
|
|
|
236 |
|
Net loss and comprehensive loss |
|
$ |
(4,162 |
) |
|
$ |
(11,846 |
) |
|
$ |
(38,426 |
) |
|
$ |
(51,507 |
) |
Net loss per share attributable to common stockholders, basic and diluted |
|
$ |
(0.08 |
) |
|
$ |
(0.24 |
) |
|
$ |
(0.74 |
) |
|
$ |
(1.03 |
) |
Weighted-average shares used in computing net loss per share attributable to common stockholders, basic and diluted |
|
|
53,082,214 |
|
|
|
50,134,104 |
|
|
|
52,148,543 |
|
|
|
50,134,104 |
|
SIGHT SCIENCES, INC.
Gross Margin Disaggregation (Unaudited)
(in thousands)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended December 31, |
|
|
Years Ended December 31, |
|
|
|
|
2025 |
|
|
2024 |
|
|
2025 |
|
|
2024 |
|
|
Revenue |
|
|
|
|
|
|
|
|
|
|
|
|
|
Interventional Glaucoma |
|
$ |
19,661 |
|
|
$ |
18,770 |
|
|
$ |
75,724 |
|
|
$ |
75,902 |
|
|
Interventional Dry Eye |
|
|
724 |
|
|
|
304 |
|
|
|
1,639 |
|
|
|
3,964 |
|
|
Total |
|
|
20,385 |
|
|
|
19,074 |
|
|
|
77,363 |
|
|
|
79,866 |
|
|
Cost of goods sold |
|
|
|
|
|
|
|
|
|
|
|
|
|
Interventional Glaucoma |
|
|
2,368 |
|
|
|
2,364 |
|
|
|
10,030 |
|
|
|
9,448 |
|
|
Interventional Dry Eye |
|
|
229 |
|
|
|
149 |
|
|
|
667 |
|
|
|
2,133 |
|
|
Total |
|
|
2,597 |
|
|
|
2,513 |
|
|
|
10,697 |
|
|
|
11,581 |
|
|
Gross profit |
|
|
|
|
|
|
|
|
|
|
|
|
|
Interventional Glaucoma |
|
|
17,293 |
|
|
|
16,406 |
|
|
|
65,694 |
|
|
|
66,454 |
|
|
Interventional Dry Eye |
|
|
495 |
|
|
|
155 |
|
|
|
972 |
|
|
|
1,831 |
|
|
Total |
|
$ |
17,788 |
|
|
$ |
16,561 |
|
|
$ |
66,666 |
|
|
$ |
68,285 |
|
|
Gross margin |
|
|
|
|
|
|
|
|
|
|
|
|
|
Interventional Glaucoma |
|
|
88.0 |
% |
|
|
87.4 |
% |
|
|
86.8 |
% |
|
|
87.6 |
% |
|
Interventional Dry Eye |
|
|
68.4 |
% |
|
|
51.0 |
% |
|
|
59.3 |
% |
|
|
46.2 |
% |
|
Total |
|
|
87.3 |
% |
|
|
86.8 |
% |
|
|
86.2 |
% |
|
|
85.5 |
% |
|

SIGHT SCIENCES, INC.
GAAP to Non-GAAP Reconciliation (Unaudited)
(in thousands)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended
December 31, |
|
|
Years Ended
December 31, |
|
|
|
|
2025 |
|
|
2024 |
|
|
2025 |
|
|
2024 |
|
|
Operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
Total Operating expenses |
|
$ |
21,457 |
|
|
$ |
28,490 |
|
|
$ |
103,765 |
|
|
$ |
118,817 |
|
|
Less: Stock-based compensation |
|
|
(2,472 |
) |
|
|
(3,915 |
) |
|
|
(12,717 |
) |
|
|
(16,763 |
) |
|
Less: Depreciation & amortization |
|
|
(108 |
) |
|
|
(176 |
) |
|
|
(487 |
) |
|
|
(712 |
) |
|
Less: Restructuring costs |
|
|
— |
|
|
|
— |
|
|
|
(2,803 |
) |
|
|
— |
|
|
Adjusted Operating Expenses(4) |
|
$ |
18,877 |
|
|
$ |
24,399 |
|
|
$ |
87,758 |
|
|
$ |
101,342 |
|
|
4 Please see section titled "Non-GAAP Financial Measures" for additional information.

SIGHT SCIENCES, INC.
Supplemental Financial Measures (Unaudited)
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended
December 31, |
|
|
|
2025 |
|
|
2024 |
|
Interventional Glaucoma active customers (5) |
|
|
1,164 |
|
|
|
1,138 |
|
Interventional Dry Eye lid treatment units sold (6) |
|
|
713 |
|
|
|
1,125 |
|
Interventional Dry Eye active customers (7) |
|
|
81 |
|
|
|
83 |
|
5 “Interventional Glaucoma active customers” means the number of customers who ordered the OMNI Surgical System or the SION Surgical Instrument during the three months ended December 31, 2025 and 2024.
6 “Interventional Dry Eye lid treatment units sold” means the quantity of TearCare SmartLids® sold during the three months ended December 31, 2025 and 2024.
7 “Interventional Dry Eye active customers” means the number of customers who ordered lid treatment units during the three months ended December 31, 2025 and 2024.

Investor Presentation March 2026

Forward-Looking Statements This presentation, together with other statements and information publicly disseminated by the Company, contains certain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, which statements are subject to considerable risks and uncertainties. The Company intends such forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in the Private Securities Litigation Reform Act of 1995. Forward-looking statements include all statements other than statements of historical fact, including statements regarding our future results of operations, product development, market opportunity, clinical trial results and timeline, and business strategy and plans. The forward-looking statements in this presentation include, but are not limited to, statements concerning the following: the Company's mission; the Company's projected financial or operational results including expectations for revenue and gross margins; estimates of the Company’s addressable markets for its products; the Company’s ability to gain share in existing markets and enter into and compete in new markets; the Company’s ability to successfully develop and commercialize its product pipeline; the Company’s ability to compete effectively; the Company’s ability to manage and grow its business, including execution of value creation initiatives; the Company's plans to invest in research and development, clinical and commercial infrastructure; the Company’s ability to successfully execute its clinical trial roadmap; the Company’s ability to successfully execute its strategic initiatives and objectives; and the Company’s ability to obtain and maintain sufficient reimbursement for its products; the Company’s expectations with respect to tariffs and other economic matters; and regulatory requirements applicable to the Company. These statements often include words such as “anticipate,” “expect,” “suggests,” “plan,” “believe,” “intend,” “estimates,” “targets,” “projects,” “should,” “could,” “would,” “may,” “will,” “forecast” and other similar expressions. Management bases these forward-looking statements on its current expectations, plans and assumptions affecting the Company’s business and industry, and such statements are based on information available to it as of the time such statements are made. Although management believes these forward-looking statements are based upon reasonable assumptions, it cannot guarantee their accuracy or completeness. Forward-looking statements are subject to and involve risks, uncertainties and assumptions that may cause the Company’s actual results, performance or achievements to be materially different from any future results, performance, or achievements predicted, assumed or implied by such forward-looking statements. Some of the risks and uncertainties that may cause actual results to materially differ from those expressed or implied by these forward-looking statements are discussed under the caption “Risk Factors” in the Company’s annual and quarterly reports with the U.S. Securities and Exchange Commission, as such may be updated from time to time in subsequent filings. These cautionary statements should not be construed by you to be exhaustive and are made only as of the date of this presentation. The Company undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by applicable law. Certain information contained in this presentation relates to, or is based on, studies, publications, surveys and other data obtained from third-party sources and the Company’s own internal estimates and research. While the Company believes these third-party sources to be reliable, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third-party sources. In addition, all of the market data included in this presentation involves a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Finally, while the Company believes its own estimates and research are reliable, such estimates and research have not been verified by any independent source. The Company has proprietary rights to trademarks, trade names and service marks appearing in this presentation that are important to its business. Solely for convenience, the trademarks, trade names and service marks may appear in this presentation without the ® and ™ symbols, but any such references are not intended to indicate that the Company forgoes or will not assert, to the fullest extent under applicable law, its rights or the rights of the applicable licensors to these trademarks, trade names and service marks. All trademarks, trade names and service marks appearing in this presentation are the property of their respective owners. The Company does not intend its use or display of other parties’ trademarks, trade names or service marks to imply, and such use or display should not be construed to imply, a relationship with, or endorsement or sponsorship of the Company by, these other parties. Without limitation, SIGHT SCIENCES™, SIGHT SCIENCES (with design)®, OMNI®, SION®, TEARCARE®, SMARTLIDS® and TruSync™ are trademarks of Sight Sciences, Inc. in the United States and other countries. RESTASIS® is a registered trademark of Allergan, Inc., and IRIS® is a registered trademark of the American Academy of Ophthalmology. Certain financial measures, including adjusted operating expenses (“non-GAAP financial measures”), were not prepared in accordance with generally accepted accounting principles in the United States (“GAAP") and are presented in this presentation to provide information that may assist investors in understanding the Company's financial and operating results. The Company believes these non-GAAP financial measures are important performance indicators because they exclude items that are unrelated to, and may not be indicative of, the Company's core financial and operating results. These non-GAAP financial measures, as calculated, may not necessarily be comparable to similarly titled measures of other companies and may not be appropriate measures for comparing the performance of other companies relative to the Company. These non-GAAP financial measures are not intended to represent, and should not be considered more meaningful measures than, or alternatives to, measures of operating performance as determined in accordance with GAAP. To the extent the Company utilizes such non-GAAP financial measures in the future, it expects to calculate them using a consistent method from period to period. Consistent with Securities and Exchange Commission regulations, the Company has not provided a reconciliation of forward-looking non-GAAP financial measures to the most directly comparable GAAP financial measures in reliance on the “unreasonable efforts” exception set forth in the applicable regulations, because there is substantial uncertainty associated with predicting any future adjustments that may be made to the Company’s GAAP financial measures in calculating the non-GAAP financial measures. For a reconciliation of non-GAAP financial measures referenced in this presentation to the most directly comparable GAAP measures, please refer to the Company's earnings release issued on March 4, 2026.

[[ Our Mission Develop transformative, interventional technologies that allow eyecare providers to procedurally elevate the standards of care — empowering people to keep seeing. | 3

A Glimpse Ahead Innovation leader in �two large, growing, underserved markets 1 TearCare market access has started with first two MACs establishing fee schedules in Oct 2025 2 Strong balance sheet in place to drive commercial growth and long-term investments 3 Strong gross margins and disciplined operating expense spend 4 The intersection of Interventional Glaucoma and Interventional Dry Eye is underway 5 | 4

INTERVENTIONAL GLAUCOMA INTERVENTIONAL DRY EYE The Intersection �of Intervention

Interventional Glaucoma
Glaucoma - Leading cause of irreversible blindness1 - Predominantly managed with daily eye drops (compliance often poor)2 Normal Mild Moderate Severe Large + Underserved Market ¹ Source: Market Scope 2025 report and JAMA Ophthalmology Prevalence of Glaucoma Among US Adults in 2022 Oct 17, 2024 . ² Newman-Casey PA, Robin AL, Blachley T, Farris KB, Heisler M, Resnicow K, Lee PP. The most common barriers to glaucoma medication adherence: A cross-sectional survey. Ophthalmology. 2015 Jul;122(7):1308-16. doi: 10.1016/j.ophtha.2015.03.026. ³ Represents Company analysis of third-party estimates in 2025. addressable U.S. market3 U.S. patients diagnosed with Glaucoma1 $6 BILLION >4 MILLION

POAG is similar to a clog in a kitchen sink: 01 TRABECULAR MESHWORK 02 SCHLEMM‘S CANAL 03 COLLECTOR CHANNELS 01 02 03 THE CONVENTIONAL OUTFLOW PATHWAY IS AN IMPORTANT FOCAL POINT IN TREATING POAG, THE MOST COMMON FORM OF GLAUCOMA. The eye’s natural drainage system is called the conventional outflow pathway. Blockage of this system prevents aqueous fluid from draining. When aqueous fluid cannot drain, intraocular pressure (IOP) rises. Elevated IOP can lead to optic nerve damage and may result in irreversible blindness. Drain Cover (trabecular meshwork): allows excess aqueous fluid to enter drainage system 01 02 Sink Pipe (Schlemm’s Canal): conducts excess aqueous fluid to exit pathways known as collector channels 03 House Plumbing (collector channels): leads excess aqueous fluid out of the eye into the venous system Primary Open-Angle Glaucoma (POAG)
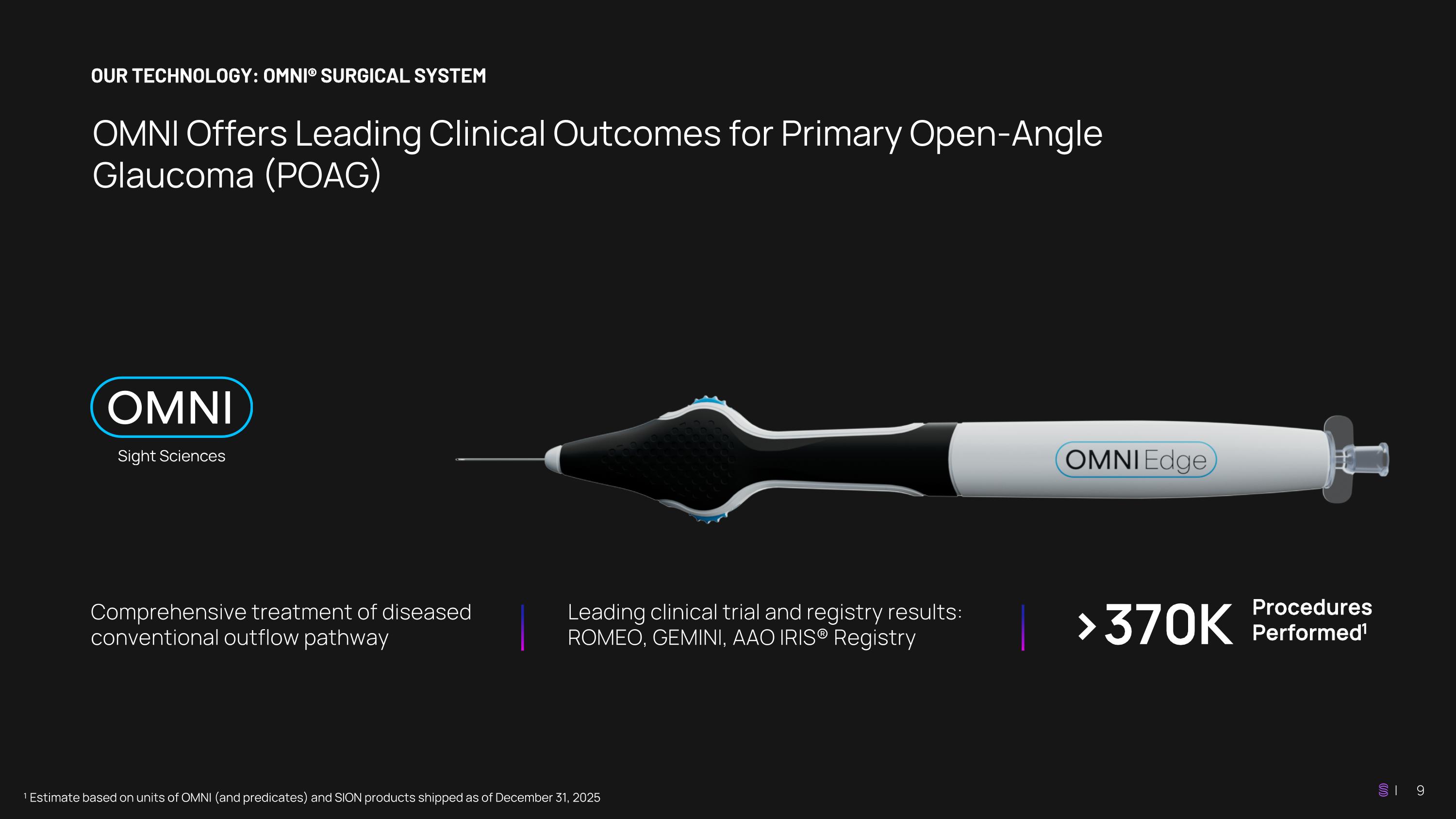
OUR TECHNOLOGY: OMNI® SURGICAL SYSTEM OMNI Offers Leading Clinical Outcomes for Primary Open-Angle Glaucoma (POAG) ¹ Estimate based on units of OMNI (and predicates) and SION products shipped as of December 31, 2025 Comprehensive treatment of diseased conventional outflow pathway Leading clinical trial and registry results: ROMEO, GEMINI, AAO IRIS® Registry >370K Procedures Performed1
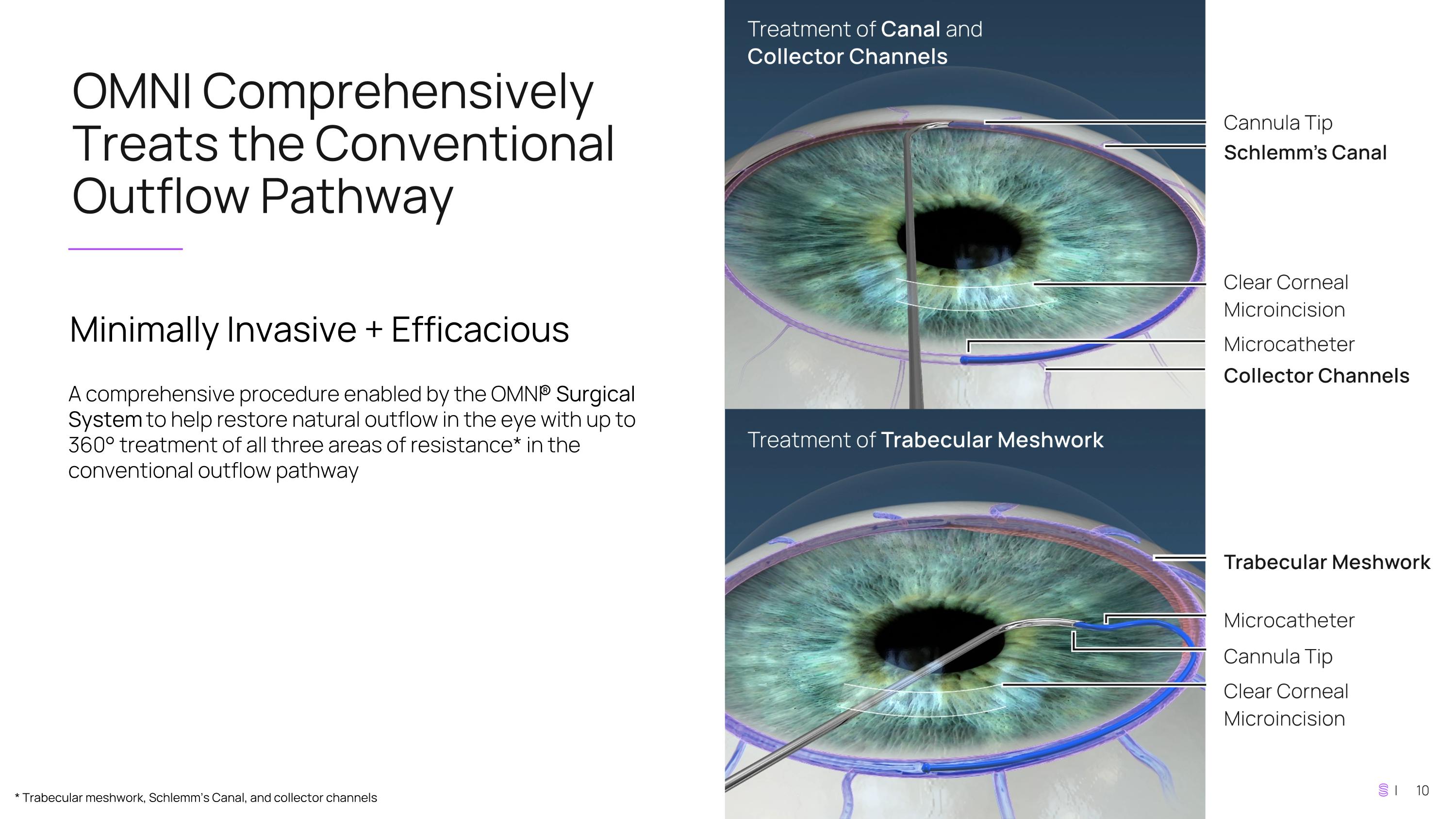
OMNI Comprehensively Treats the Conventional Outflow Pathway Minimally Invasive + Efficacious A comprehensive procedure enabled by the OMNI® Surgical System to help restore natural outflow in the eye with up to 360° treatment of all three areas of resistance* in the conventional outflow pathway * Trabecular meshwork, Schlemm’s Canal, and collector channels

OMNI is Proven with Robust Clinical Evidence & Broad �FDA Indication AGIS-7 Findings3 <18 mmHg is the target IOP to limit the progression of glaucoma. On average, there was zero change in visual field defect score for patients whose IOP stayed below 18 mmHg over 6 years. OMNI is the most comprehensive implant-free Minimally Invasive Glaucoma Surgery (MIGS) technology, designed to effectively treat the full spectrum of primary open-angle glaucoma (POAG)1 OMNI with patented TruSync™ Technology is the only MIGS device with an FDA indication that allows for: Use in combination cataract or standalone (without cataract) procedures Access to 360 degrees of the diseased conventional outflow pathway through a clear corneal microincision Comprehensive treatment of all three areas of resistance2 in the diseased conventional outflow pathway Use in adult patients with POAG across the spectrum of �disease severity 1 Dickerson J, et al. Ab Interno Canaloplasty and Trabeculotomy Outcomes for Mild, Moderate, and Advanced Open-Angle Glaucoma: A ROMEO Analysis. Clin Ophthalmol. 2024: 18 1433-1440. �2 Trabecular meshwork, Schlemm’s Canal, and collector channels 3 The Advanced Glaucoma Intervention Study (AGIS): 7. The relationship between control of intraocular pressure and visual field deterioration. The AGIS Investigators 4 GEMINI 36-month paper (Greenwood MR, Yadgarov A, Flowers BE, Sarkisian SR, Ohene-Nyako A, Dickerson JE Jr. 36-month outcomes from the prospective GEMINI study: canaloplasty and trabeculotomy combined with cataract surgery for patients with primary open-angle glaucoma. Clin Ophthalmol 2023;17:3817-3824.) 4
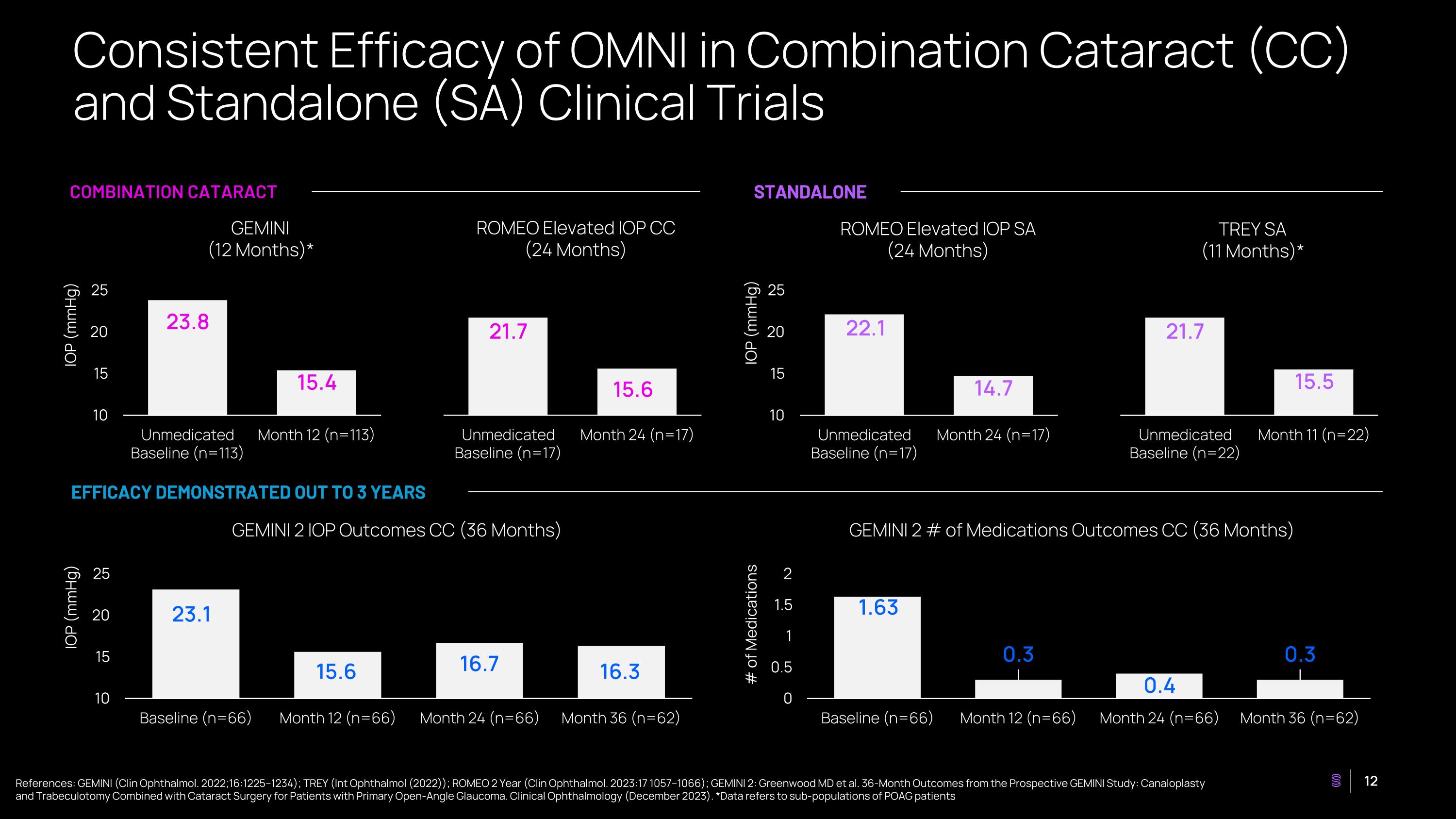
Consistent Efficacy of OMNI in Combination Cataract (CC) and Standalone (SA) Clinical Trials COMBINATION CATARACT STANDALONE EFFICACY DEMONSTRATED OUT TO 3 YEARS GEMINI (12 Months)* ROMEO Elevated IOP CC (24 Months) ROMEO Elevated IOP SA (24 Months) TREY SA (11 Months)* GEMINI 2 IOP Outcomes CC (36 Months) GEMINI 2 # of Medications Outcomes CC (36 Months) IOP (mmHg) IOP (mmHg) IOP (mmHg) # of Medications References: GEMINI (Clin Ophthalmol. 2022;16:1225–1234); TREY (Int Ophthalmol (2022)); ROMEO 2 Year (Clin Ophthalmol. 2023:17 1057–1066); GEMINI 2: Greenwood MD et al. 36-Month Outcomes from the Prospective GEMINI Study: Canaloplasty and Trabeculotomy Combined with Cataract Surgery for Patients with Primary Open-Angle Glaucoma. Clinical Ophthalmology (December 2023). *Data refers to sub-populations of POAG patients

OMNI Addresses All Six MIGS POAG Categories and Allows Surgeons to Customize Treatment ¹ Represents Company analysis of third-party estimates based on 2025 data MILD DISEASE (40%) MODERATE DISEASE (40%) ADVANCED DISEASE (20%) ~$2B opportunity ~$2B opportunity ~$1B opportunity ~$0.4B opportunity ~$0.4B opportunity ~$0.2B opportunity $5B Opportunity1 $1B Opportunity1 STANDALONE MIGS ~85%1 of POAG Eyes COMBINATION CATARACT MIGS ~15%1 of POAG Eyes MARKET OPPORTUNITY1

Large and Unmet Clinical Need for Standalone MIGS Combination Cataract Standalone | 14 ~15% of POAG eyes1, ~90% of MIGS procedures1 Established, growing market Benefits from inherent IOP-lowering effect of cataract surgery Share-taking driven by efficacy, fast recovery times and attractive safety profile ~85% of POAG eyes¹, ~10% of MIGS procedures1 Large, underserved patient population MIGS procedure is the SOLE reason for operating room visit Standalone adoption requires a procedure with robust safety and efficacy, without the benefit of cataract surgery ¹ Represents Company analysis of third-party estimates based on 2025 data.
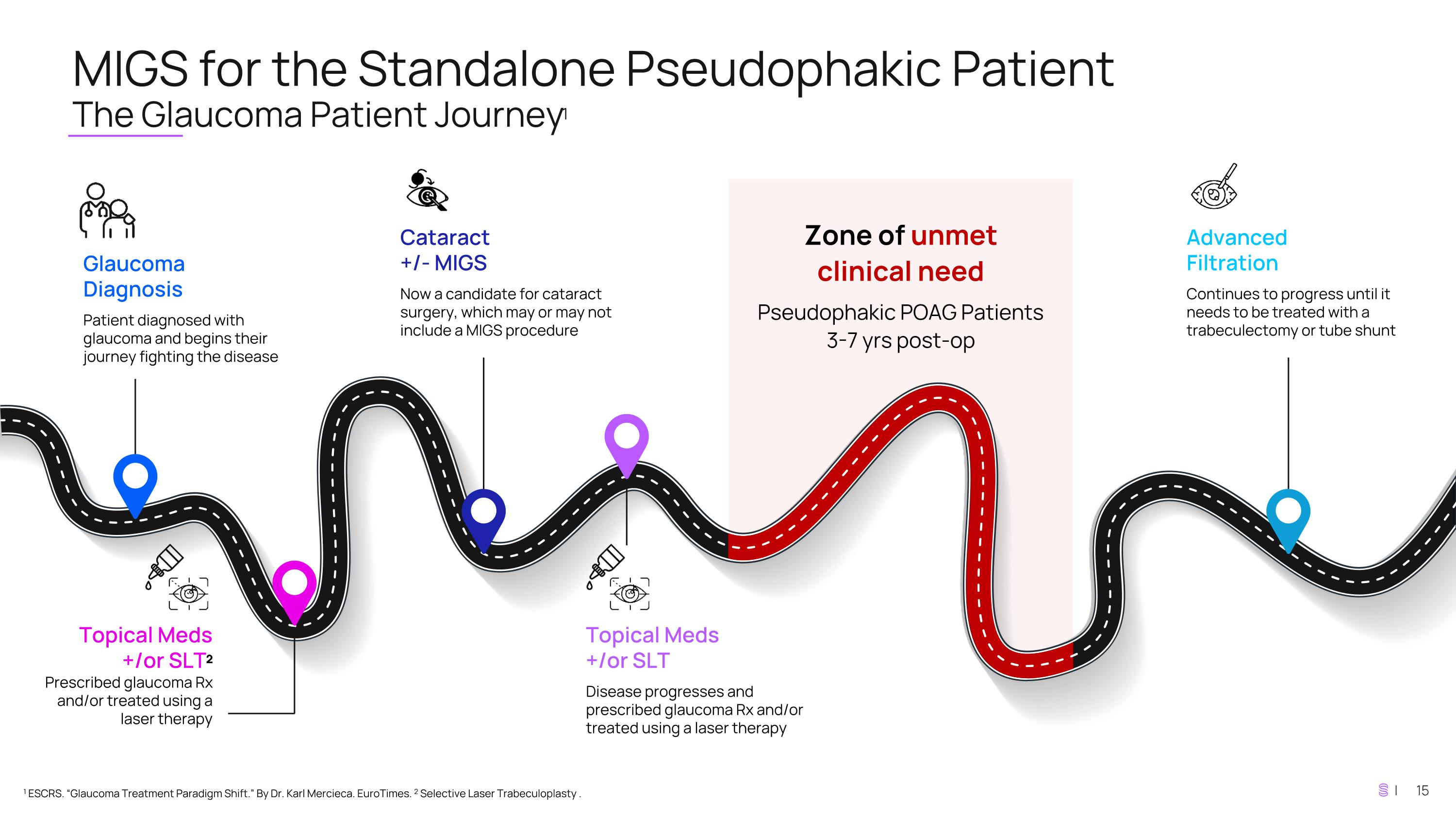
MIGS for the Standalone Pseudophakic Patient �The Glaucoma Patient Journey1 1 ESCRS. “Glaucoma Treatment Paradigm Shift.” By Dr. Karl Mercieca. EuroTimes. 2 Selective Laser Trabeculoplasty . Zone of unmet �clinical need Pseudophakic POAG Patients �3-7 yrs post-op Glaucoma Diagnosis Patient diagnosed with glaucoma and begins their journey fighting the disease Topical Meds +/or SLT2 Prescribed glaucoma Rx and/or treated using a laser therapy Cataract�+/- MIGS Now a candidate for cataract surgery, which may or may not include a MIGS procedure Topical Meds �+/or SLT Disease progresses and prescribed glaucoma Rx and/or treated using a laser therapy Advanced �Filtration Continues to progress until it needs to be treated with a trabeculectomy or tube shunt
Interventional Dry Eye
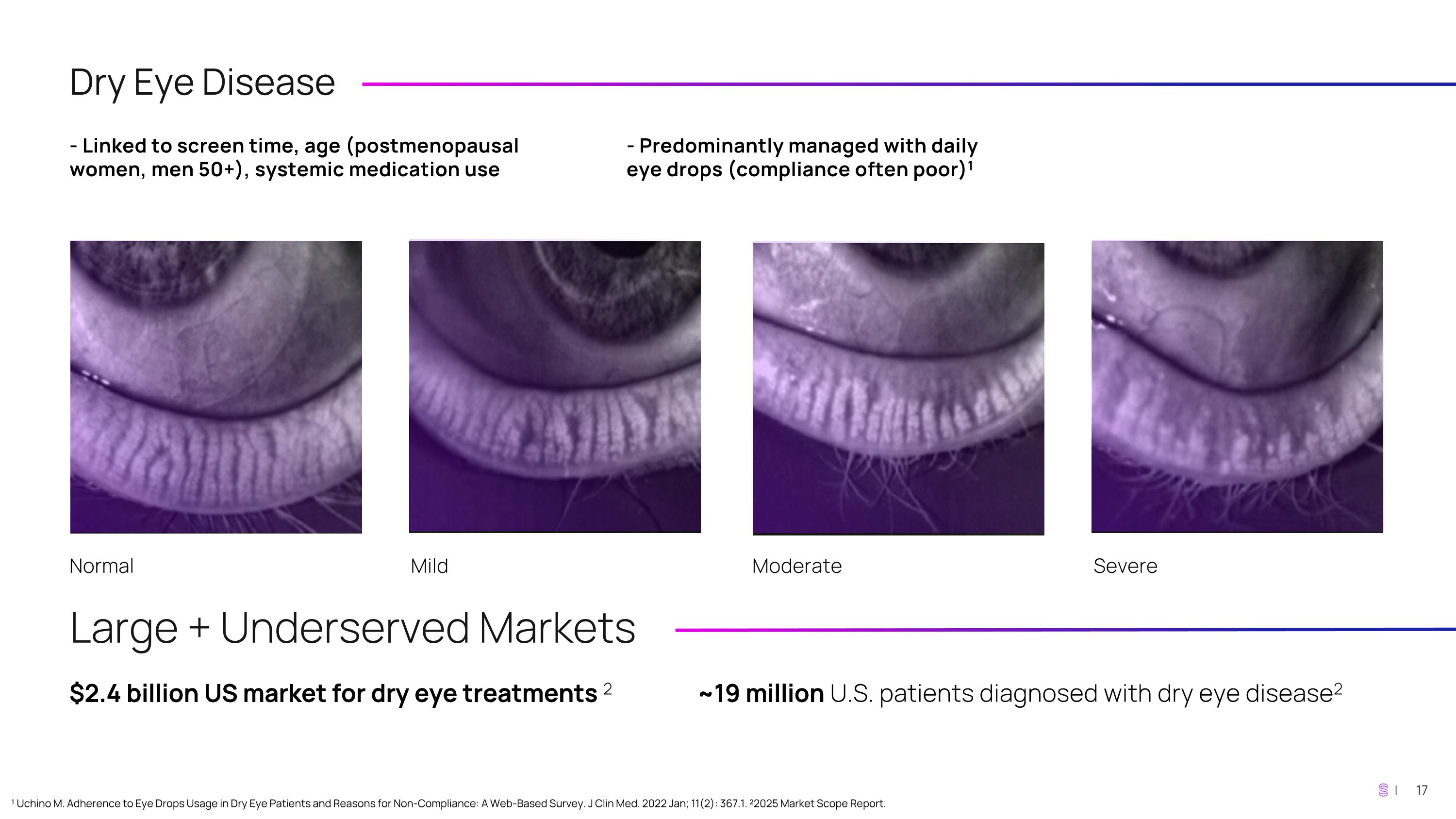
Dry Eye Disease - Linked to screen time, age (postmenopausal women, men 50+), systemic medication use - Predominantly managed with daily eye drops (compliance often poor)1 Normal Mild Moderate Severe Large + Underserved Markets $2.4 billion US market for dry eye treatments 2 ~19 million U.S. patients diagnosed with dry eye disease2 ¹ Uchino M. Adherence to Eye Drops Usage in Dry Eye Patients and Reasons for Non-Compliance: A Web-Based Survey. J Clin Med. 2022 Jan; 11(2): 367.1. ²2025 Market Scope Report.
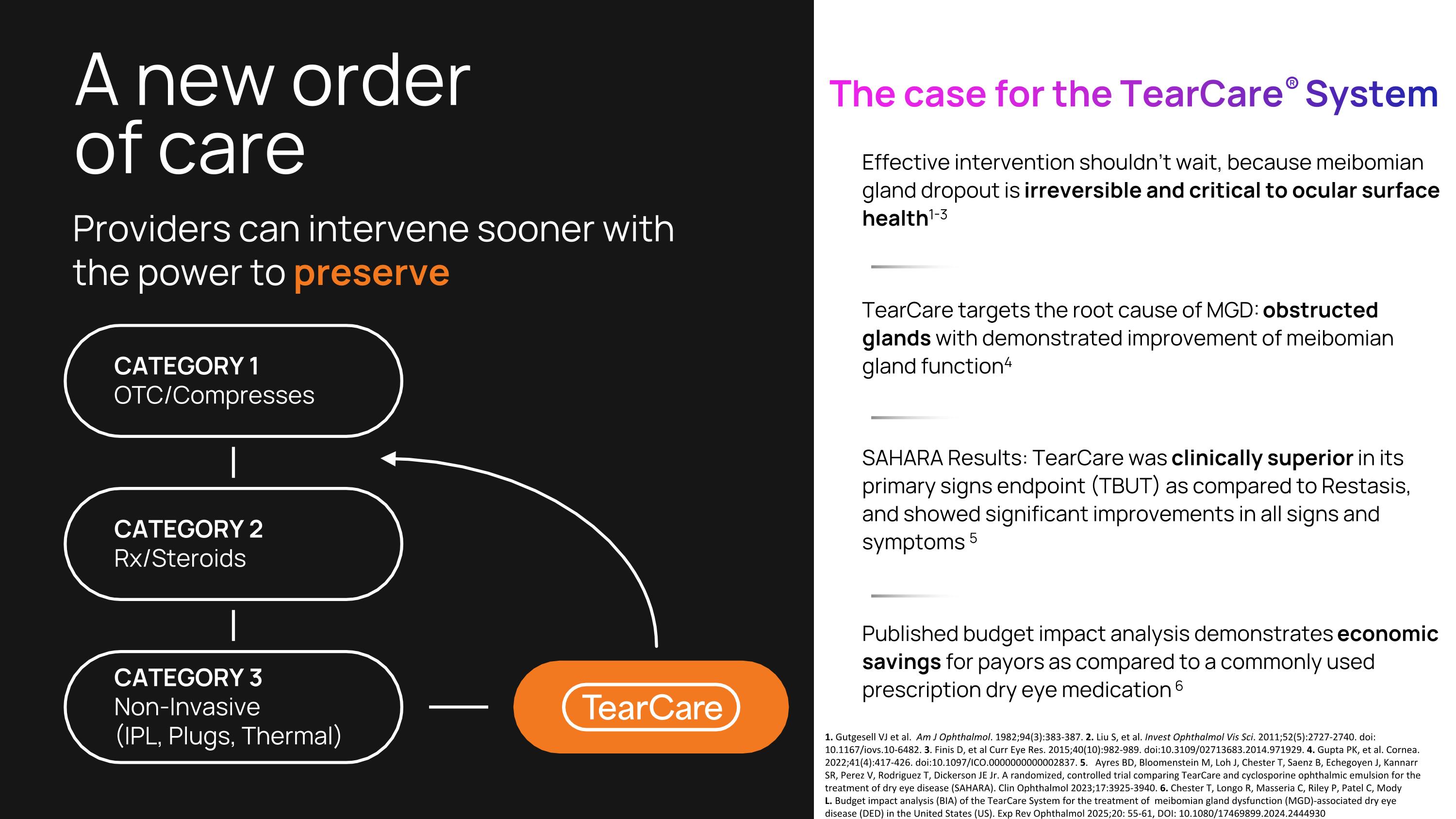
A new order �of care Effective intervention shouldn’t wait, because meibomian gland dropout is irreversible and critical to ocular surface health1-3 TearCare targets the root cause of MGD: obstructed glands with demonstrated improvement of meibomian gland function4 SAHARA Results: TearCare was clinically superior in its primary signs endpoint (TBUT) as compared to Restasis, and showed significant improvements in all signs and symptoms 5 Published budget impact analysis demonstrates economic savings for payors as compared to a commonly used prescription dry eye medication 6 Providers can intervene sooner with the power to preserve The case for the TearCare® System CATEGORY 1 OTC/Compresses CATEGORY 2 Rx/Steroids CATEGORY 3 Non-Invasive �(IPL, Plugs, Thermal) 1. Gutgesell VJ et al. Am J Ophthalmol. 1982;94(3):383-387. 2. Liu S, et al. Invest Ophthalmol Vis Sci. 2011;52(5):2727-2740. doi: 10.1167/iovs.10-6482. 3. Finis D, et al Curr Eye Res. 2015;40(10):982-989. doi:10.3109/02713683.2014.971929. 4. Gupta PK, et al. Cornea. 2022;41(4):417-426. doi:10.1097/ICO.0000000000002837. 5. Ayres BD, Bloomenstein M, Loh J, Chester T, Saenz B, Echegoyen J, Kannarr SR, Perez V, Rodriguez T, Dickerson JE Jr. A randomized, controlled trial comparing TearCare and cyclosporine ophthalmic emulsion for the treatment of dry eye disease (SAHARA). Clin Ophthalmol 2023;17:3925-3940. 6. Chester T, Longo R, Masseria C, Riley P, Patel C, Mody L. Budget impact analysis (BIA) of the TearCare System for the treatment of meibomian gland dysfunction (MGD)-associated dry eye disease (DED) in the United States (US). Exp Rev Ophthalmol 2025;20: 55-61, DOI: 10.1080/17469899.2024.2444930

MGD Opportunity U.S. patients diagnosed with Dry Eye Disease (DED) 1 Million DED patients ~19 Up to 86% of DED is associated with poor tear quality due to Meibomian Gland Dysfunction (MGD )1, 2 Million MGD patients ~13 – 16 ~50% of DED patients have moderate to severe symptoms 1 (most likely to seek treatment + targeted patient population in SAHARA RCT) Million moderate to severe MGD DED patients ~7 – 8 ¹ Market Scope 2025 Dry Eye Device Report. ² Lemp MA, Crews LA, Bron AJ, Foulks GN, Sullivan BD. Distribution of aqueous-deficient and evaporative dry eye in a clinic-based patient cohort: a retrospective study. Cornea. 2012;31(5):472-478.

MGD is an Underserved Disease State The current market is dominated by eyedrops that do not address the underlying causes of MGD 1 Many dry eye treatments focus on increasing tear volume in aqueous deficient patients No interventional standard of care for treatment of MGD There is poor patient compliance with the use of Rx and OTC eyedrops for treatment 2 The US market for dry eye treatments was $2.4 billion in 2025 1 ¹ Market Scope 2025 Dry Eye Device Report and Dry Eye Pharmaceuticals Report and internal estimates. ² Uchino M. Adherence to Eye Drops Usage in Dry Eye Patients and Reasons for Non-Compliance: A Web-Based Survey. J Clin Med. 2022 Jan; 11(2): 367.1.
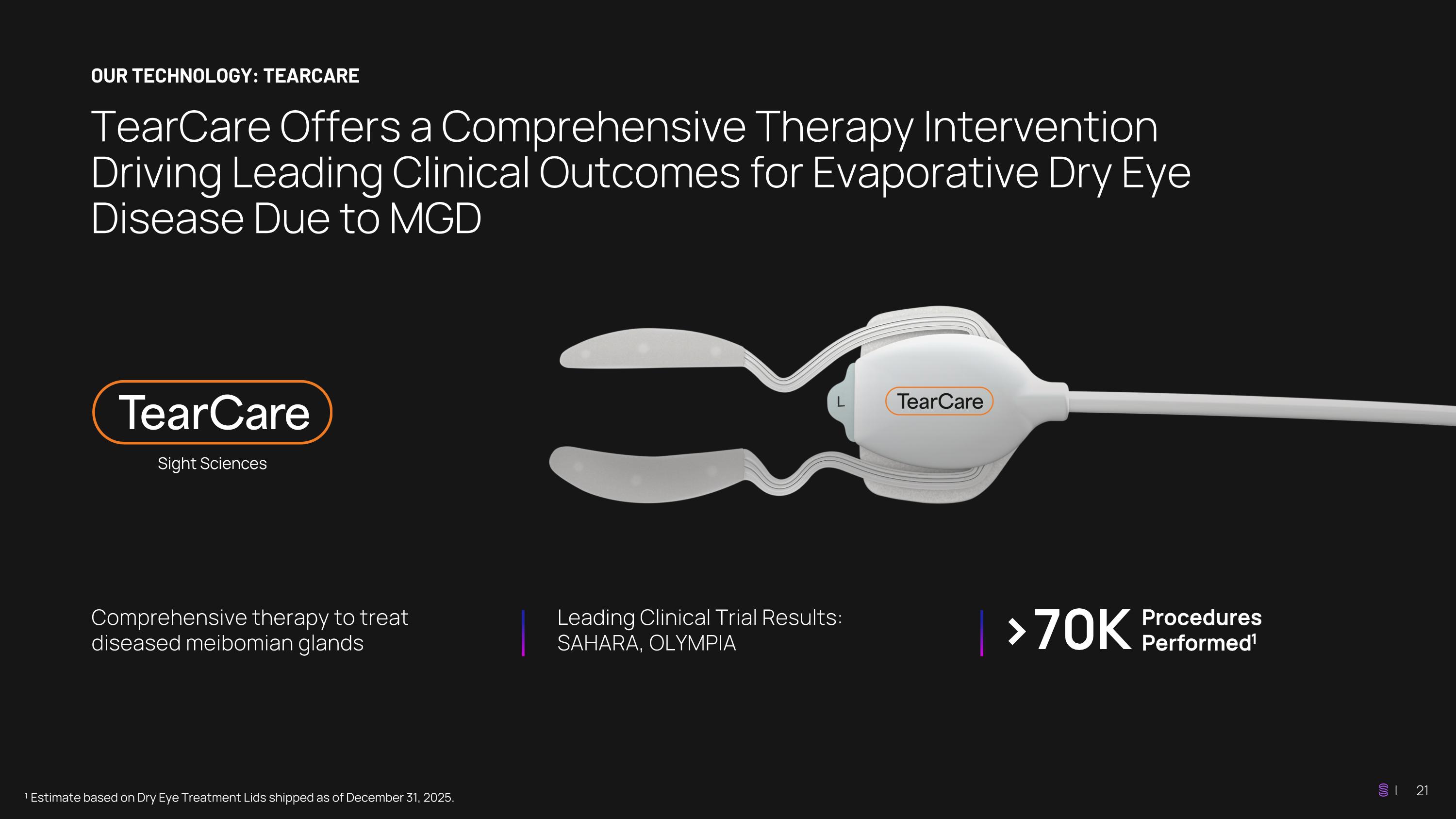
TearCare Offers a Comprehensive Therapy Intervention Driving Leading Clinical Outcomes for Evaporative Dry Eye Disease Due to MGD OUR TECHNOLOGY: TEARCARE ¹ Estimate based on Dry Eye Treatment Lids shipped as of December 31, 2025. Comprehensive therapy to treat diseased meibomian glands Leading Clinical Trial Results: SAHARA, OLYMPIA >70K Procedures Performed1

TearCare: Designed to Preserve and Improve Gland Functionality TearCare is the only FDA-cleared interventional, open-eye, thermal-activated gland expression therapy designed to treat MGD conveniently and comfortably ¹ Gupta et al. Cornea 2022;41:417–426 Thin, wearable SmartLids® conform to the eyelid and allow natural blinking Precise, consistent, software- controlled thermal therapeutic melting cycle (at 45°C +/- 0.7°C for 15 minutes)¹ Comprehensive gland clearing protocol allows providers to manually evacuate the melted meibum comfortably 01 Application 02 Therapy 03 Expression

SAHARA RCT Randomized Controlled Trial comparing TearCare and Restasis®1 Signs Superiority + Durability 2 + Head-to-Head Study TearCare vs Restasis1 + Large Trial (N=345) + Randomized + Assessor Masked + 3 Stages + Long-term (2-year trial) ¹ Restasis is a trademark of Allergan™ an AbbVie company ² Endpoints for SAHARA include superiority over Restasis at six months in our primary objective endpoint, tear break-up time. Study through 24 months to show duration of effectiveness. Ayres BD, Bloomenstein MR, Loh J, et al. A Randomized, Controlled Trial Comparing TearCare® and Cyclosporine Ophthalmic Emulsion for the Treatment of Dry Eye Disease (SAHARA). Clin Ophthalmol. 2023;17:3925-3940. Hovanesian J, Ayres BD, Bloomenstein MR, Loh J, Chester T, Saenz B, Echegoyen J, Kannarr SR, Rodriguez TC, Dickerson JE Jr. Durability of the TearCare treatment effect in subjects with dry eye disease: Stage 3 of the Sahara randomized controlled trial. Optom Vision Sci 2025;102:495-504 doi:10.1097/OPX.0000000000002278.
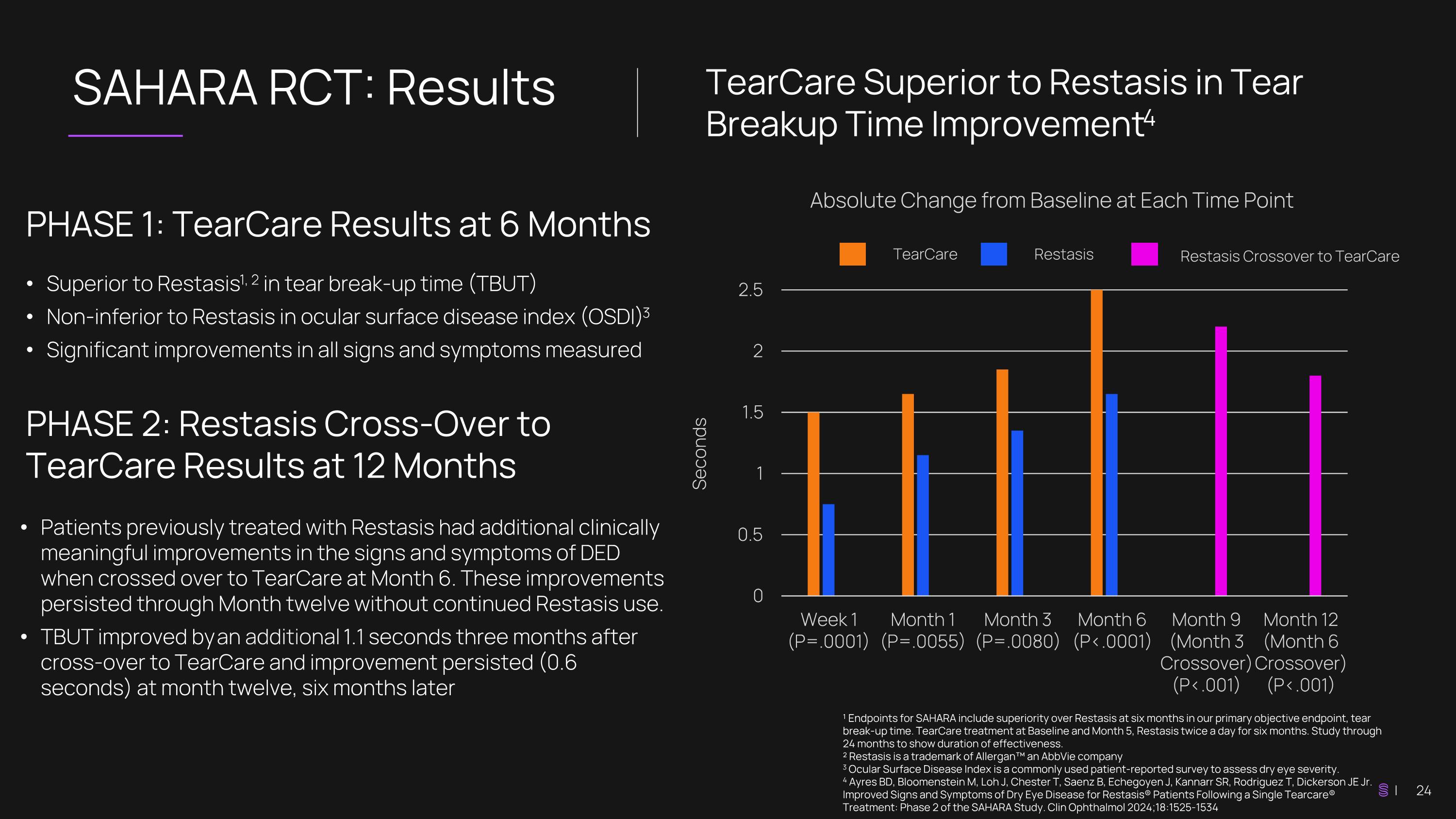
SAHARA RCT: Results ¹ Endpoints for SAHARA include superiority over Restasis at six months in our primary objective endpoint, tear break-up time. TearCare treatment at Baseline and Month 5, Restasis twice a day for six months. Study through 24 months to show duration of effectiveness. ² Restasis is a trademark of Allergan™ an AbbVie company 3 Ocular Surface Disease Index is a commonly used patient-reported survey to assess dry eye severity. 4 Ayres BD, Bloomenstein M, Loh J, Chester T, Saenz B, Echegoyen J, Kannarr SR, Rodriguez T, Dickerson JE Jr. Improved Signs and Symptoms of Dry Eye Disease for Restasis® Patients Following a Single Tearcare® Treatment: Phase 2 of the SAHARA Study. Clin Ophthalmol 2024;18:1525-1534 TearCare Superior to Restasis in Tear Breakup Time Improvement4 PHASE 1: TearCare Results at 6 Months Superior to Restasis1, 2 in tear break-up time (TBUT) Non-inferior to Restasis in ocular surface disease index (OSDI) 3 Significant improvements in all signs and symptoms measured Seconds Absolute Change from Baseline at Each Time Point TearCare Restasis Patients previously treated with Restasis had additional clinically meaningful improvements in the signs and symptoms of DED when crossed over to TearCare at Month 6. These improvements persisted through Month twelve without continued Restasis use. TBUT improved by an additional 1.1 seconds three months after cross-over to TearCare and improvement persisted (0.6 seconds) at month twelve, six months later PHASE 2: Restasis Cross-Over to TearCare Results at 12 Months Restasis Crossover to TearCare
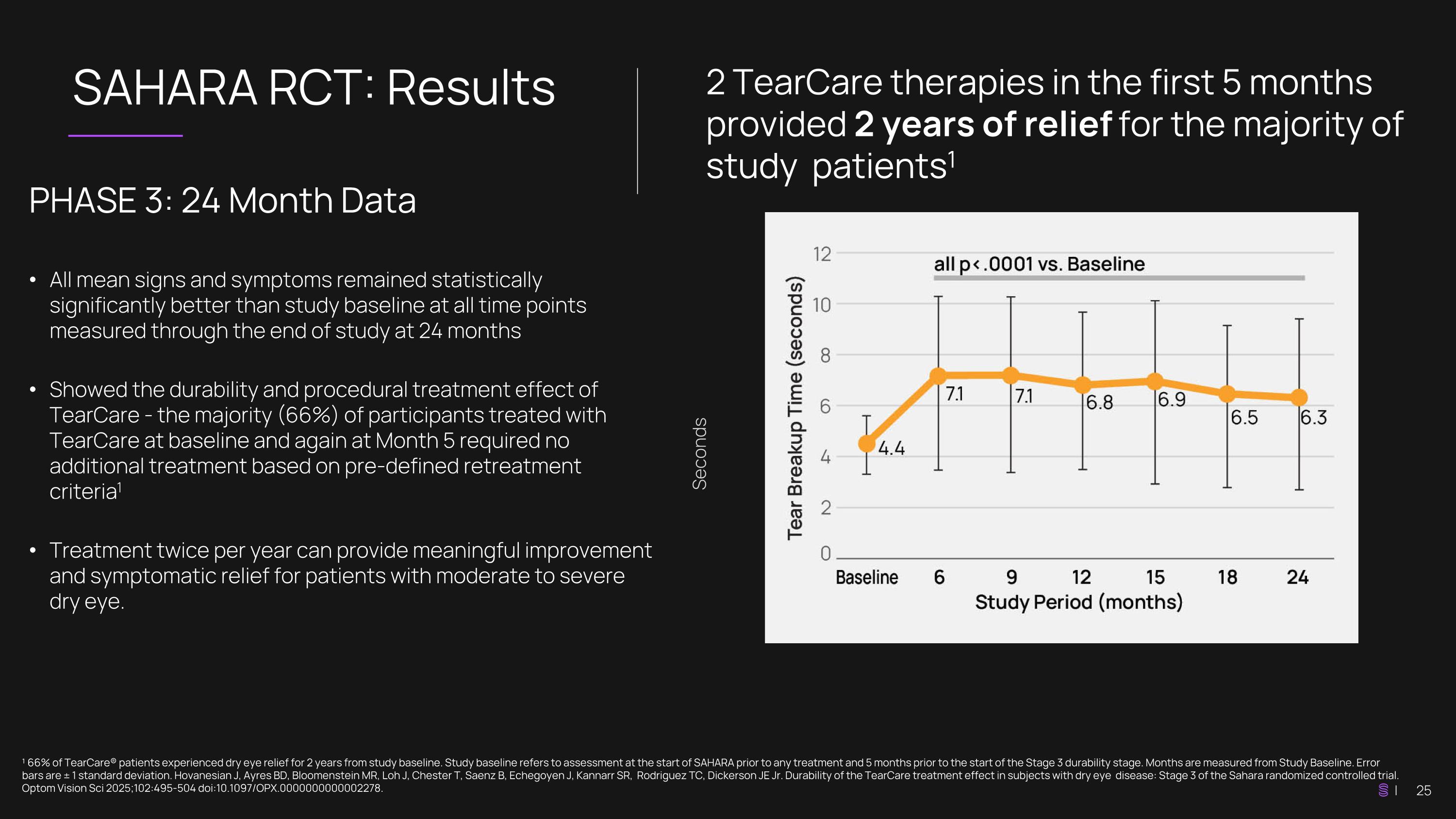
SAHARA RCT: Results ¹ 66% of TearCare® patients experienced dry eye relief for 2 years from study baseline. Study baseline refers to assessment at the start of SAHARA prior to any treatment and 5 months prior to the start of the Stage 3 durability stage. Months are measured from Study Baseline. Error bars are ± 1 standard deviation. Hovanesian J, Ayres BD, Bloomenstein MR, Loh J, Chester T, Saenz B, Echegoyen J, Kannarr SR, Rodriguez TC, Dickerson JE Jr. Durability of the TearCare treatment effect in subjects with dry eye disease: Stage 3 of the Sahara randomized controlled trial. Optom Vision Sci 2025;102:495-504 doi:10.1097/OPX.0000000000002278. 2 TearCare therapies in the first 5 months provided 2 years of relief for the majority of study patients1 PHASE 3: 24 Month Data All mean signs and symptoms remained statistically significantly better than study baseline at all time points measured through the end of study at 24 months Showed the durability and procedural treatment effect of TearCare - the majority (66%) of participants treated with TearCare at baseline and again at Month 5 required no additional treatment based on pre-defined retreatment criteria1 Treatment twice per year can provide meaningful improvement and symptomatic relief for patients with moderate to severe dry eye. Seconds

Interventional Dry Eye Strategy:�Targeted + Scalable Growth ¹ Estimated as of June 30, 2025 based on review of claims data and Company analytics 2 As of December 31, 2025 Scaling Commercially While Actively Pursuing Expanded Market Access With the power of TearCare, we can: Improve the lives of U.S. MGD patients Scale commercial resources with market access wins Target ~6,500 physicians identified as most likely to adopt MGD treatment procedures¹ Activate a large installed customer base, over 70,000 SmartLids sold2, built across real-world testing and data collection since 2019 Leverage synergies with our Interventional Glaucoma customer base and commercial infrastructure
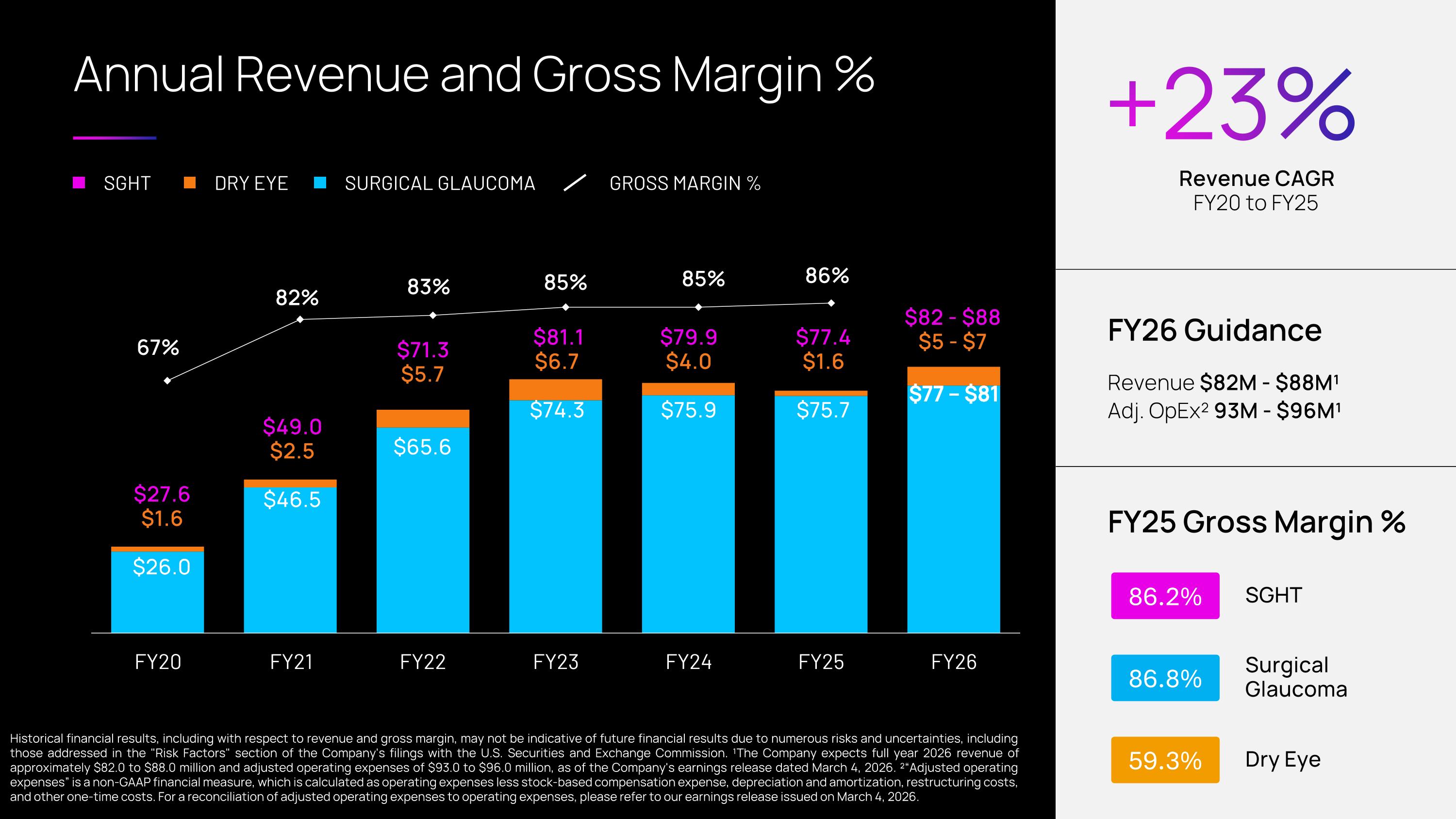
$82 - $88 $5 - $7 Annual Revenue and Gross Margin % SGHT DRY EYE SURGICAL GLAUCOMA GROSS MARGIN % $27.6 $1.6 $26.0 $49.0 $2.5 $46.5 $71.3 $5.7 $65.6 $81.1 $6.7 $74.3 $79.9 $4.0 $75.9 +23% FY25 Gross Margin % Revenue CAGR FY20 to FY25 FY26 Guidance Revenue $82M - $88M¹ Adj. OpEx² 93M - $96M¹ 86.2% 86.8% 59.3% SGHT Surgical �Glaucoma Dry Eye Historical financial results, including with respect to revenue and gross margin, may not be indicative of future financial results due to numerous risks and uncertainties, including those addressed in the "Risk Factors" section of the Company's filings with the U.S. Securities and Exchange Commission. ¹The Company expects full year 2026 revenue of approximately $82.0 to $88.0 million and adjusted operating expenses of $93.0 to $96.0 million, as of the Company's earnings release dated March 4, 2026. ²“Adjusted operating expenses” is a non-GAAP financial measure, which is calculated as operating expenses less stock-based compensation expense, depreciation and amortization, restructuring costs, and other one-time costs. For a reconciliation of adjusted operating expenses to operating expenses, please refer to our earnings release issued on March 4, 2026. $77.4 $1.6 $75.7

Keep Seeing TM





























